CS1 rationale
What CS1 is and why it matters
CS1 is Cereno Scientific’s lead clinical-stage drug candidate, developed as an oral, once-daily add-on treatment for Pulmonary Arterial Hypertension (PAH).
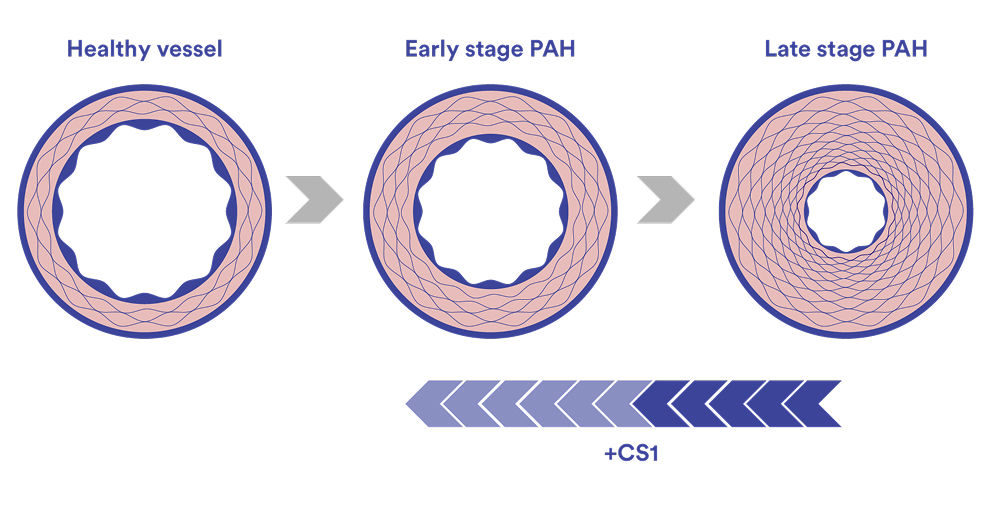
PAH is a rare and progressive disease in which changes to the pulmonary blood vessels increase pressure in the lungs and place growing strain on the right side of the heart. While current therapies improve symptoms and outcomes, they largely focus on vasodilation and often do not address the structural and biological changes that drive disease progression.
CS1 is designed to complement existing PAH therapies by targeting key disease-driving processes — with the ambition of offering disease-modifying potential combined with favorable safety and tolerability suitable for long-term use.
CS1 has received Orphan Drug Designation in both the United States and Europe, reflecting the significant unmet medical need in PAH and supporting its continued development.
Fast Track designation from the U.S. FDA, enabling closer regulatory interaction and supporting an efficient development process.

Designed for combination treatment in real-world PAH care
PAH is commonly treated using combination therapy, where patients receive multiple drugs that act on different pathways. CS1 is intended to be used as an add-on therapy, not as a replacement for existing treatments.
This positioning is deliberate:
- CS1 targets disease biology not directly addressed by current vasodilator therapies
- It is designed to integrate into existing treatment regimens
- Once-daily oral dosing supports convenience and adherence in chronic care
By fitting naturally into current treatment strategies, CS1 aims to be clinically relevant from both a physician and patient perspective.
Scientific rationale: addressing disease progression, not just symptoms
The progression of PAH is driven by a combination of:
-
Abnormal vascular remodeling – thickening and stiffening of pulmonary arteries
-
Fibrosis – scarring of vessel walls and surrounding tissue
-
Inflammation – contributing to ongoing vascular damage
-
Right-heart dysfunction – resulting from sustained pressure overload
CS1 builds on Cereno’s pioneering work in epigenetic modulation through histone deacetylase (HDAC) inhibition, an approach designed to influence how genes involved in these processes are regulated.
Rather than acting on a single downstream pathway, HDAC inhibition has the potential to influence multiple interconnected mechanisms involved in PAH progression.
Treatment aim
CS1 has the potential to stop, halt or reverse the PAH disease progression
PAH is characterized by thickening and narrowing of the small arteries in the lungs, including the development of characteristic plexiform lesions, which restrict blood flow from the right side of the heart to the lungs. Over time, these changes combined with increased tissue scarring (fibrosis), reduce the elasticity of the blood vessels and increase resistance to blood flow. This process, known as vascular remodeling, raises the pressure in the pulmonary arteries and impairs circulation. The disease-modifying effects of CS1 has the potential to stop, halt or reverse the PAH disease progression by reverse vascular remodeling.
Formulation and treatment concept
CS1 is a proprietary, controlled-release formulation developed specifically for use in cardiovascular disease.
Key features include:
- Once-daily oral administration
- Sustained exposure over 24 hours
- A formulation strategy designed for chronic treatment
- Development informed by the well-characterised safety profile of the active compound
This differentiates CS1 from several existing PAH therapies that require frequent dosing, inhalation, infusion, or complex administration, which can add burden for patients over time.

Understanding the disease
Pulmonary Arterial Hypertension (PAH)
Pulmonary Arterial Hypertension is a rare, progressive condition caused by narrowing and structural changes in the pulmonary arteries. Over time, these changes increase pulmonary pressure and impair right-heart function.
Despite treatment advances, PAH remains associated with significant morbidity and reduced life expectancy, highlighting the need for new therapeutic approaches that go beyond symptom control.
Clinical evidence
CS1 Phase IIa trial in PAH
A Phase IIa trial evaluating the safety, tolerability pharmacokinetics, and exploratory efficacy of CS1 on top of standard therapy in patients with PAH was completed in 2024. The Phase IIa trial was conducted at 10 US clinics over 12 weeks with a total of 25 patients of which 21 were evaluated for efficacy parameters. The trial successfully met its primary endpoint of safety and tolerability, with no drug-related serious adverse events.
The exploratory Phase IIa trial of CS1 identified efficacy signals suggesting reversal of pathological remodeling of pulmonary vessels. This was observed through:
- Signals of improved right ventricular function, which is the most significant predictor of mortality in PAH was observed through improvement of right ventricular global longitudinal strain (RV GLS) and reduced tricuspid regurgitation (TR)
- Signals of improved overall cardiac function was observed through improved NYHA/WHO functional class and Quality of Life (QoL)
- Signals of disease modification and prognosis was observed through improved REVEAL 2.0 risk score

Clinical evidence
Expanded Access Program for CS1 in PAH
Upon request from patients and physicians, an Expanded Access Program (EAP) was initiated as an extension of the Phase IIa trial in PAH. The EAP enables Cereno to collect long term safety and tolerability as well as insights on efficacy data on CS1 use in PAH patients under an FDA approved protocol. The study met primary endpoint of safety and tolerability after 12-months. The EAP plays an important role in strengthening the clinical package for CS1 by providing additional long-term data beyond the Phase IIa trial.
A sub-study was initiated in February 2025 to obtain further insights and visualization of long-term treatment of CS1 on top of standard therapy. The innovative imaging technology used is called Functional Respiratory Imaging (FRI), developed by Fluidda, is a non-invasive tool to provide detailed, patient-specific insights into pulmonary vascular changes that may provide valuable insights into CS1’s disease-modifying potential. The program will run for 12 months.
Further analysis of the EAP including results from the Fluidda imaging sub-study will be communicated during the second quarter of 2026.
Read our policy on EAP further down.

Next steps
Global multicenter - and placebocontrolled phase IIb study
The clinical development plan for CS1 is focused on continuing to evaluate it as a well-tolerated, orally administered therapy with a favorable safety profile and robust disease-modifying effects in PAH. Building on the promising results from the completed Phase IIa trial, a larger, placebo-controlled Phase IIb trial is currently being planned. The upcoming Phase IIb trial is designed to further evaluate the safety, tolerability and efficacy of different doses of CS1. The new global, multicenter, placebo-controlled trial will be conducted in collaboration with a leading international contract research organization (CRO).
The Phase IIb trial is planned to begin during June 2026.

Why CS1 stands out
These attributes position CS1 as a differentiated and potentially meaningful addition to the evolving PAH treatment landscape:
- Targets underlying disease mechanisms, not only symptoms
- Designed for add-on use in combination therapy
- Once-daily oral dosing for patient convenience
- Favorable safety and tolerability observed to date
- Supported by early clinical signals, regulatory recognition, and orphan status
Expanded access policy
At Cereno Scientific, our mission is to develop pioneering treatments to enhance and extend life for people living with rare cardiovascular and pulmonary diseases. We recognize the urgent needs faced by patients and their families, and we are committed to bring safer and more effective treatments to patients with high unmet needs.
We understand that there are times when an individual is unable to participate in a clinical trial, and other treatment options have been exhausted. In those cases, the individual's physician may choose to request access to an investigational drug outside of a clinical trial via what is often termed “Expanded Access” in the United States.
Expanded access, sometimes called “compassionate use” in the US, allows patients with serious or life-threatening conditions to gain access to investigational treatments outside of clinical trials, when no comparable alternatives exist. While participation in clinical trials remains the preferred path to access, we understand that this may not always be possible. If you are interested to learn more about expanded access, please visit the US FDA’s website.
Cereno Scientific's Expanded Access Program that is for individual patients who have completed the CS1 Phase IIa trial in PAH (NCT06321705) has ended.
Cereno Scientific does not have active Expanded Access Programs at this time.
In line with the 21st Century Cures Act, Cereno Scientific may revise this policy at any time.

